We felt that cars were one of the biggest culprits when it came to carbon dioxide emission, especially since approximately 25% of cars are causing 90% of the pollution in our environment. Considering that the number of cars in the world is continuously increasing, and the CO₂ emissions continue to increase, we are likely to be strongly affected by global warming and air pollution.
In order to be able to implement the idea of adding a catalyst to a car, we felt it important to research into the current car engine to make sure our ideas could work as part of it, as well as improve rather than hinder its efficiency.
-
Cylinder
-
The Four Strokes
-
Car Engine
-
Catalytic Converter
<
>
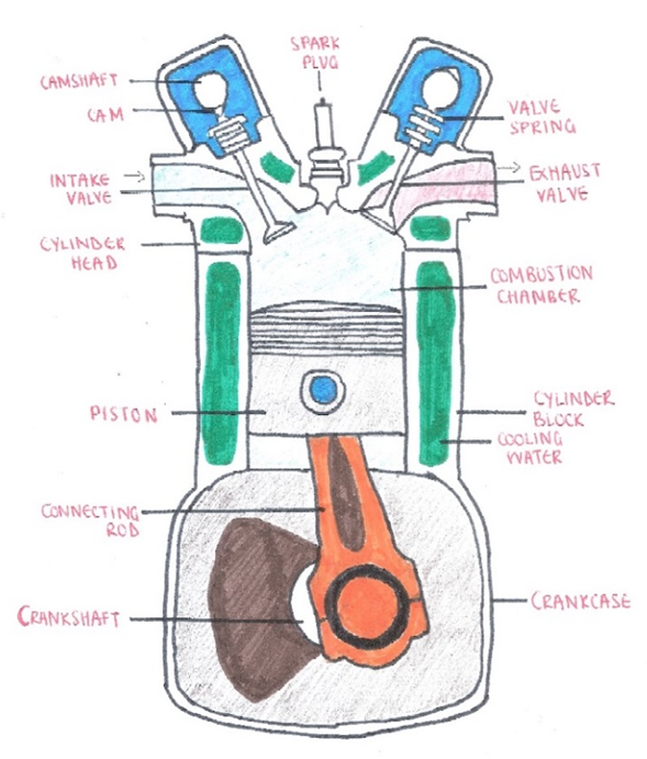
In this day and age we’ve come to take cars for granted; they are designed to allow us to move from one place to another without having to rely on public transport like trains or slower means of transport like the horse and cart of a century earlier. A car engine is able to convert chemical energy from fuels to kinetic energy that allows cars to travel for hundreds of miles on a full tank. This energy conversion is carried out in cylinders inside the engine, and consists of four stages in the four-stroke engine.
Intake stroke: the intake valve opens, while the piston moves down at the same time to draw in a mixture of air and fuel from the carburettor, which is responsible for mixing the air and fuel. This works because there is a difference in atmospheric pressure between outside the combustion chamber and inside. The exhaust valve remains closed.
Compression stroke: in this stroke, the intake valve first closes so that the air and fuel mixture inside cannot escape. The piston moves up in order to compress the mixture inside the combustion chamber, thereby increasing the chance of ignition.
Power stroke: once the piston has reached the top of its stroke, and the air and fuel mixture is at the maximum amount of compression, a spark from the spark plug is emitted, igniting the fuel-air mixture. Internal combustion occurs, causing the fuel to burn and rapidly expand, pushing the piston down again. The spark itself is caused by a high voltage electrical charge produced in the ignition coil, which travels through the distributor and into the spark plug.
Exhaust stroke: the exhaust valve opens to let the waste gases out. The momentum from the combustion allows the piston to move up again, which forces these wastes gases out. At the end of the exhaust stroke, the exhaust valve closes while the intake valve opens again, and the cycle repeats itself.
Compression stroke: in this stroke, the intake valve first closes so that the air and fuel mixture inside cannot escape. The piston moves up in order to compress the mixture inside the combustion chamber, thereby increasing the chance of ignition.
Power stroke: once the piston has reached the top of its stroke, and the air and fuel mixture is at the maximum amount of compression, a spark from the spark plug is emitted, igniting the fuel-air mixture. Internal combustion occurs, causing the fuel to burn and rapidly expand, pushing the piston down again. The spark itself is caused by a high voltage electrical charge produced in the ignition coil, which travels through the distributor and into the spark plug.
Exhaust stroke: the exhaust valve opens to let the waste gases out. The momentum from the combustion allows the piston to move up again, which forces these wastes gases out. At the end of the exhaust stroke, the exhaust valve closes while the intake valve opens again, and the cycle repeats itself.
A typical car engine consists of four, six or eight cylinders, which can be arranged in different ways depending on the type of car they are found in.Each of the pistons is connected to a connecting rod which is connected to a crankshaft that turns in order for the pistons to move up and down. Movement of the crankshaft during three of the four strokes is maintained by the momentum of the flywheel and the crank itself. Usually on top of the cylinders is the camshaft, which coordinates the opening and closing of the intake and exhaust valves. The movements of the crankshaft and camshaft are synchronised by the timing belt which is connected to both, and this ensures that fuel enters each cylinder at the correct stroke and waste gases leave at the correct stroke. The alternator is responsible for generating electric power, which is used to charge the car battery.
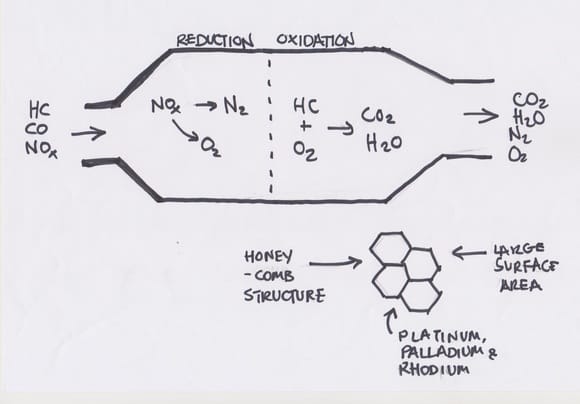
One of the drawbacks of conventional cars that use fossil fuels is that the incomplete combustion process can release poisonous and generally harmful gases. The incomplete combustion of hydrocarbons can result in carbon monoxide being produced, which is very toxic inside the body and may result in suffocation inside the body. Along with carbon monoxide, very high temperatures in the cylinder means that nitrogen and oxygen from the air may react together to produce nitrous oxides, which can cause problems like causing inflammation in the respiratory system and making plants more vulnerable to disease. Unreacted hydrocarbons could also be released, which could cause hydrocarbon poisoning. These problems can be avoided by adding a catalytic converter to cars, which is responsible for converting these more harmful substances to less harmful ones.
The metal casing of catalytic converters contains a honeycomb structure made of ceramic and coated with catalysts. The most effective catalytic converters make use of two different catalysts to catalyse the reactions, and are called three-way catalytic converters because they tackle three substances – carbon monoxide, nitrous oxides and unburned hydrocarbons. The catalysts themselves are made up of mixtures of platinum, palladium and rhodium, all three of which are precious metals, and are therefore very costly.
Catalysts
One of the catalysts used in the catalytic converter is made up of platinum and rhodium and is called the reduction catalyst. This is because it is responsible for separating oxygen molecules from nitrogen molecules, which results in two harmless gases, nitrogen and oxygen, being formed and released from the tailpipe instead of nitrous oxides.
The oxidation catalyst is made up of platinum and palladium and is used to convert the carbon monoxide and unburned hydrocarbons to less harmful substances. In these reactions, the carbon monoxide and hydrocarbons are reacted with oxygen that was not used up in the cylinder; this is also known as oxidation. Carbon dioxide is the main by-product of this reaction, with the oxidation of hydrocarbons causing water vapour to also be formed.
The oxidation catalyst is made up of platinum and palladium and is used to convert the carbon monoxide and unburned hydrocarbons to less harmful substances. In these reactions, the carbon monoxide and hydrocarbons are reacted with oxygen that was not used up in the cylinder; this is also known as oxidation. Carbon dioxide is the main by-product of this reaction, with the oxidation of hydrocarbons causing water vapour to also be formed.