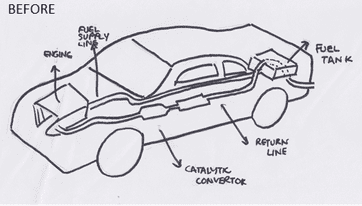
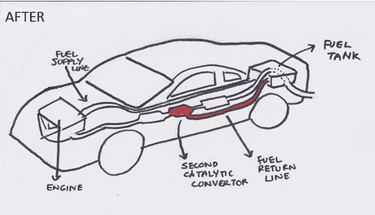
A car already has one catalytic convertor, turning toxic gases into carbon dioxide and nitrogen. Our idea as a group is to use a second catalyst to convert carbon dioxide into ethanol, a fuel that can be used by the car.
The basis of our idea, a catalyst that converts CO2 in to fuel, had already been perfected by scientists. Our solution is to input this catalyst into cars to reduce carbon emissions that would affect both human health and the environment. As this was the case, along with the fact nanoparticles would be used, we were aware we could not test materials for the catalyst. However, we still wanted to carry out some sort of experimentation to demonstrate we understood the principles of the catalyst that was discovered.
We set out to resemble their findings by catalysing the decomposition of hydrogen peroxide and see which substance works best as a catalyst and these were our results.
We set out to resemble their findings by catalysing the decomposition of hydrogen peroxide and see which substance works best as a catalyst and these were our results.
From the results that we observed, it was evident that manganese (IV) oxide was the best catalyst in this experiment, as it decomposed the hydrogen peroxide the quickest. This can be seen by the amount of foam that was produced. Some of the other substances used as catalysts were less effective, but still worked as we observed fizzing in the measuring cylinder.
The Catalyst
|
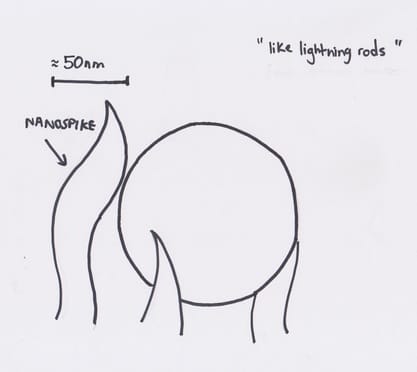
The proposed catalyst is made up of carbon, copper and nitrogen and “its novelty lies in its nanoscale structure, consisting of copper nanoparticles embedded in carbon spikes.” These carbon spikes are around 50 nm in height and are likened to “tiny lightning rods”. They are so small that it “concentrates electrochemical reactivity at the tip of the spike” which would not occur with a normal catalyst which is flat. Two advantages of this catalyst would be that it runs the entire reaction itself and that the production of ethanol is at a yield around 65% which is quite large.
This diagram shows how the catalyst would fit in the car. The fuel that could be produced through this new catalyst could be siphoned off by an extra pipe and injected back into the fuel chamber, as shown above.
.
Properties of the New Converter
Liquid Crystal Polymers- The slight problem with the nanospike catalyst is that it may require the carbon dioxide to be dissolved in an aqueous solution, like water, however water would eventually evaporate due to the heat. A solution is to use a liquid crystal polymer as they are unreactive and can withstand high temperatures. Another possible solution would be to use a solid catalyst in the form of a honeycomb structure that current catalytic converters use as this creates a large surface area for reactions.
Materials- The catalyst uses copper, carbon, and nitrogen, all three of which are largely abundant, which means we are using less rare materials to complete the job, making our idea sustainable.
Ethanol- The main porduct of the reaction within the catalyst is ethanol, which means that engines do not have to be re-adapted, as current engines are already adapted to do so.
Environmentally Friendly- Our idea allows one to travel a lot further on the same tank of fuel, but at t he same time, helps reduce carbon emissions. It's also sustainable because consumers can still use current petrol stations to fill up and it helps reduce our use of fossil fuels.
Materials- The catalyst uses copper, carbon, and nitrogen, all three of which are largely abundant, which means we are using less rare materials to complete the job, making our idea sustainable.
Ethanol- The main porduct of the reaction within the catalyst is ethanol, which means that engines do not have to be re-adapted, as current engines are already adapted to do so.
Environmentally Friendly- Our idea allows one to travel a lot further on the same tank of fuel, but at t he same time, helps reduce carbon emissions. It's also sustainable because consumers can still use current petrol stations to fill up and it helps reduce our use of fossil fuels.